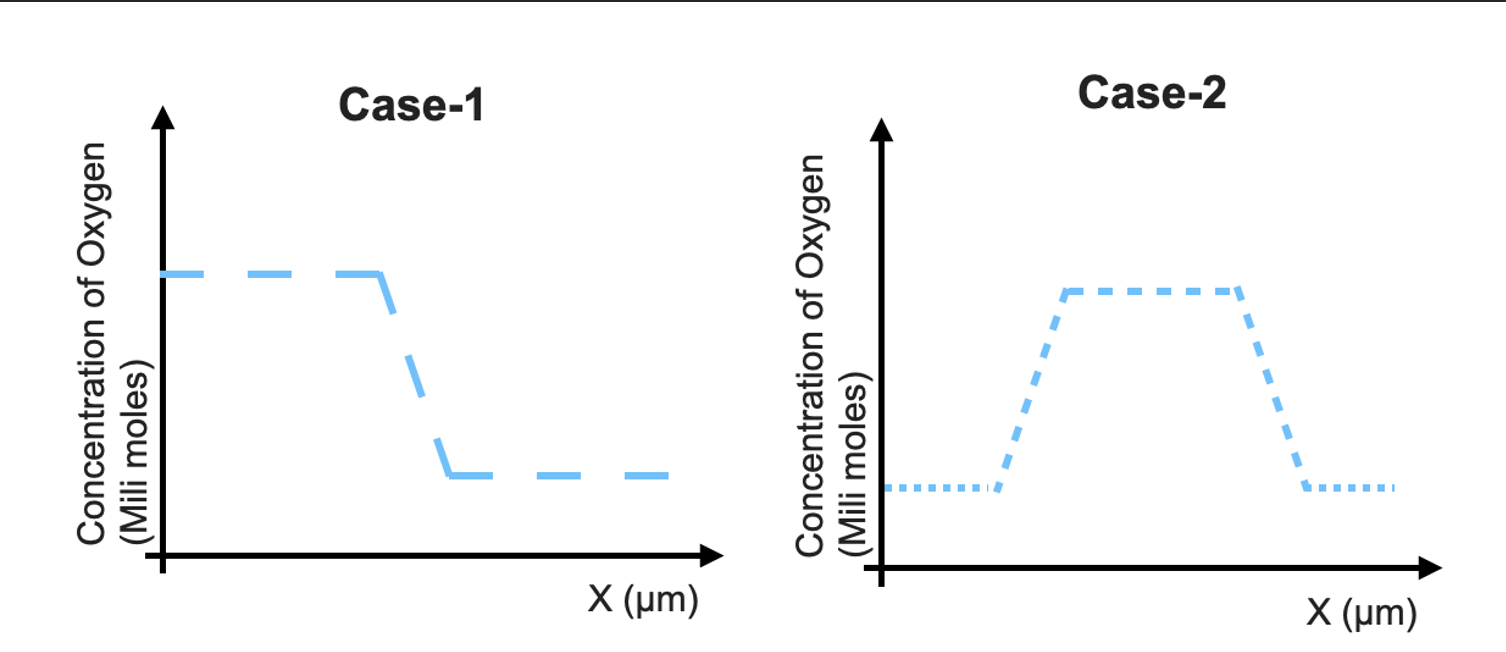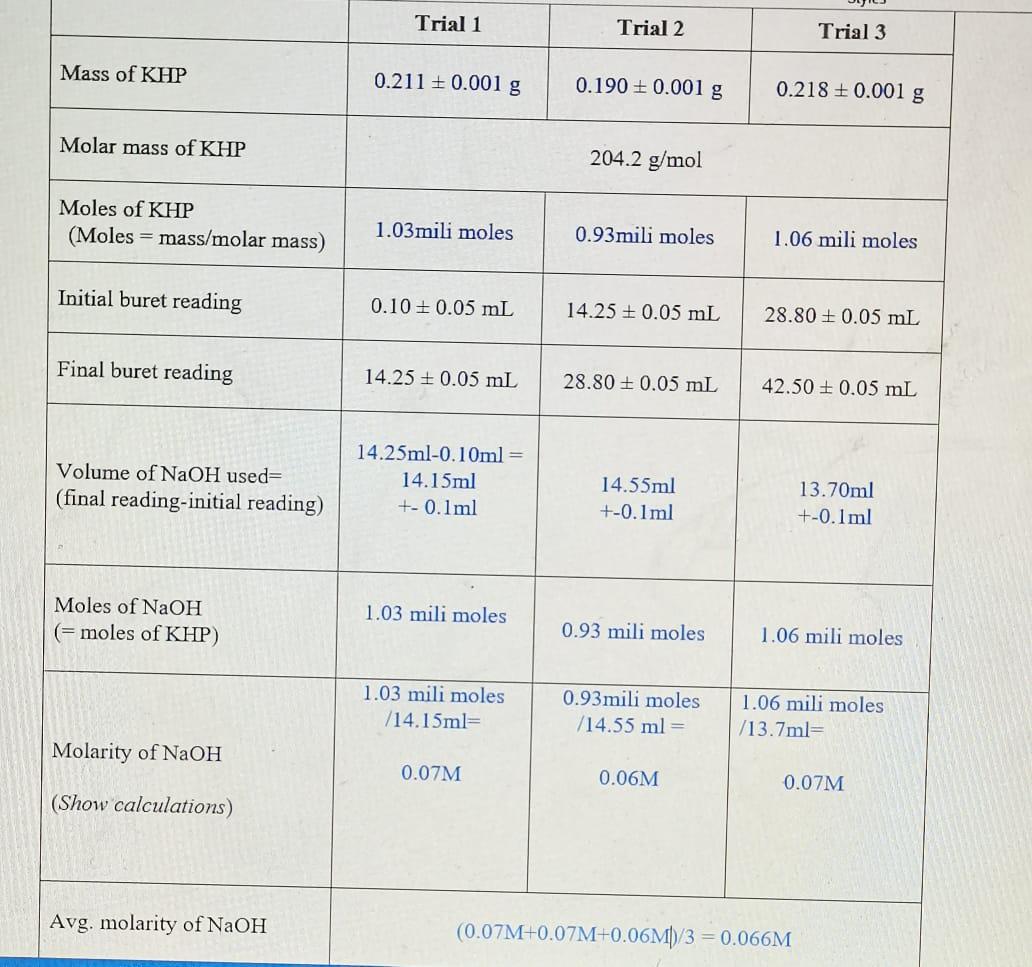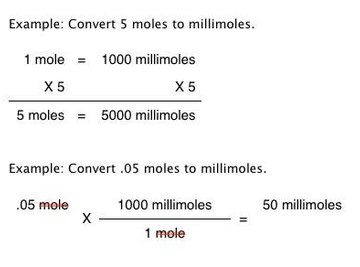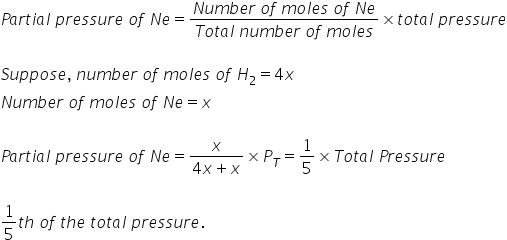
an ideal gas mixture contains 4 milimoles of H2 for every mili mole of Ne .The partial pressure of Ne is
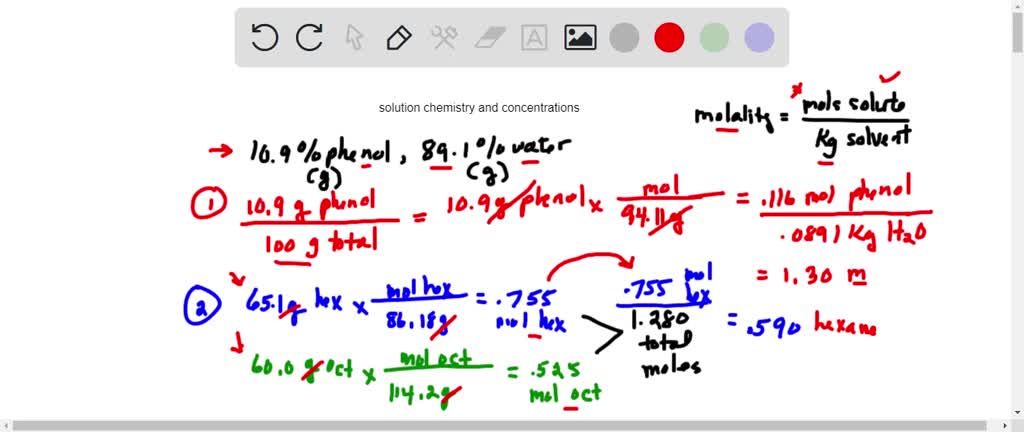
SOLVED: The percent by mass of phenol (MM = 94.11 glmol) in an aqueous solution is 10.9%. What is the molality of the phenol solution? 0 of 1 point earned 3 attempts

How many milli moles of sucrose should be dissolved in 500 gms of water so that the difference between the boiling point and freezing point of the solution becomes 103.57^oC ? (Kf =

The pHof a buffersolution prepared by adding 10 mLof 0.1 M CH_(3) COOH and 20 mL 0.1 M sodium ac... - YouTube

Variation of the amount of ammonium and sodium chloride exchanged in... | Download Scientific Diagram

SOLVED: The percent by mass of phenol (MM = 94.11 glmol) in an aqueous solution is 10.9%. What is the molality of the phenol solution? 0 of 1 point earned 3 attempts